In the third period of the table, the atoms all have a neon-like core of 10 electrons, and shell #3 is occupied progressively with eight electrons, starting with the 3s-orbital. As we progress from lithium (atomic number=3) to neon (atomic number=10) across the second row or period of the table, all these atoms start with a filled 1s-orbital, and the 2s-orbital is occupied with an electron pair before the 2p-orbitals are filled. Shell #2 has four higher energy orbitals, the 2s-orbital being lower in energy than the three 2p-orbitals. According to the Aufbau principle, the electrons of an atom occupy quantum levels or orbitals starting from the lowest energy level, and proceeding to the highest, with each orbital holding a maximum of two paired electrons (opposite spins).Įlectron shell #1 has the lowest energy and its s-orbital is the first to be filled.
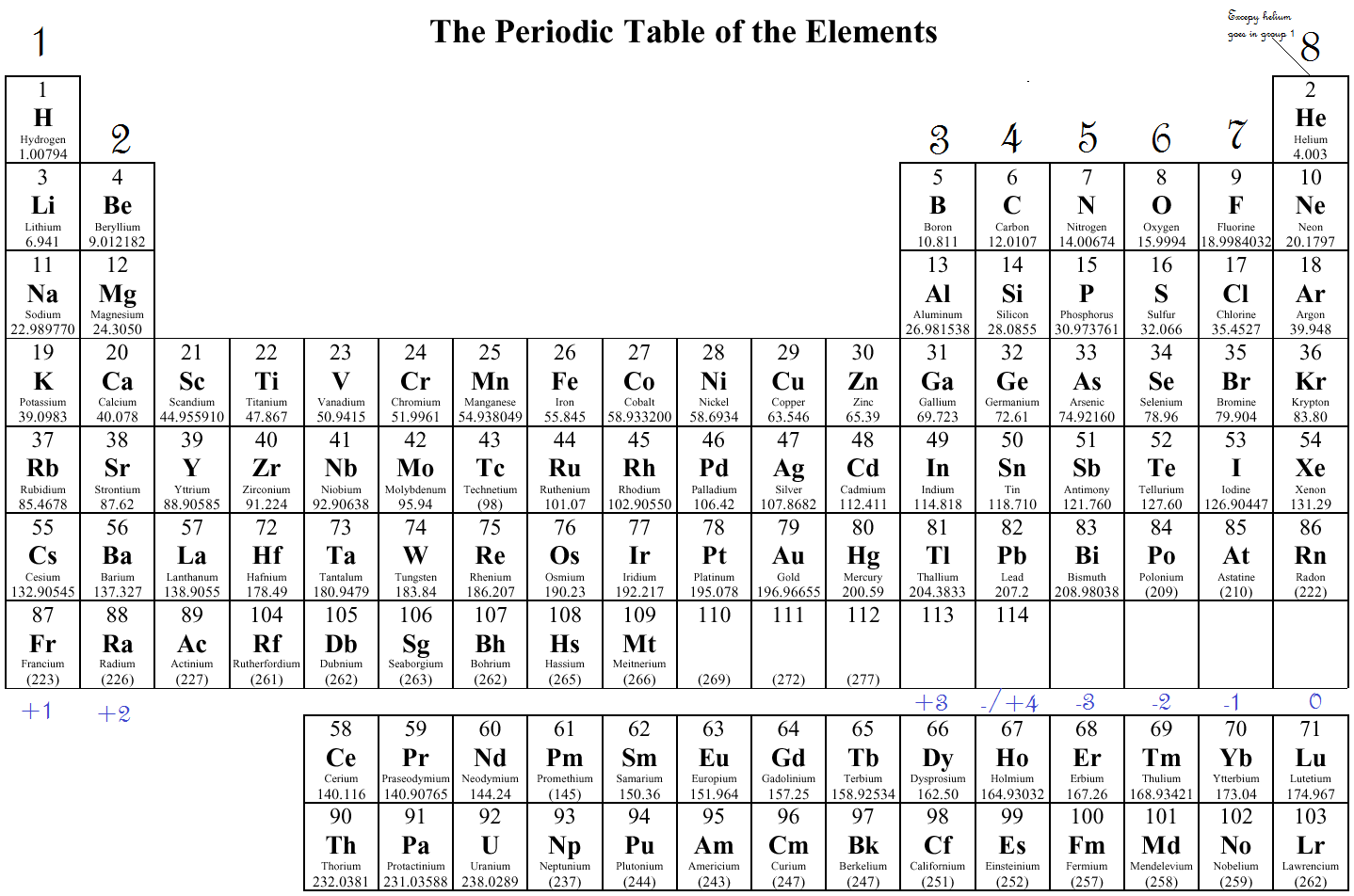
The truncated periodic table shown above provides the orbital electronic structure for the first eighteen elements (hydrogen through argon).

Consequently, our understanding of organic chemistry must have, as a foundation, an appreciation of the electronic structure and properties of these elements. Other interactive periodic tables provide comprehensive data for each element, including nuclide properties, environmental and health factors, presentation in different languages and much more.įor comic relief you may wish to examine a periodic table linked to element references in comic books.įour elements, hydrogen, carbon, oxygen and nitrogen, are the major components of most organic compounds. There are, of course, over eighty other elements.Ī complete periodic table, having very useful interactive links has been created by Mark Winter. The periodic table shown here is severely truncated. This module introduces some basic facts and principles that are needed for a discussion of organic molecules.Įlectron Configurations in the Periodic Table 1A The study of organic chemistry must at some point extend to the molecular level, for the physical and chemical properties of a substance are ultimately explained in terms of the structure and bonding of molecules. Okay, so for chlorine, we see that we have our seven valence electrons in the outer shell, with all the remaining electrons serving as our inner core electrons.Electron Configurations & The Periodic Table And in this third show we have our seven valence electrons, The remaining 10 electrons are in shells too, And one they total up to a total of electrons. And you can see here with this representation of the chlorine atom We have here are 3rd shell, so and equals three. So that would mean out of the 17 total electrons. Chlorine has an atomic number of 17 When it is neutral it has 17 protons. So you're gonna say total electrons Which is connected to your atomic number for neutral element equals your valence electrons plus your inner core electrons. We're going to say here that your total number of electrons.
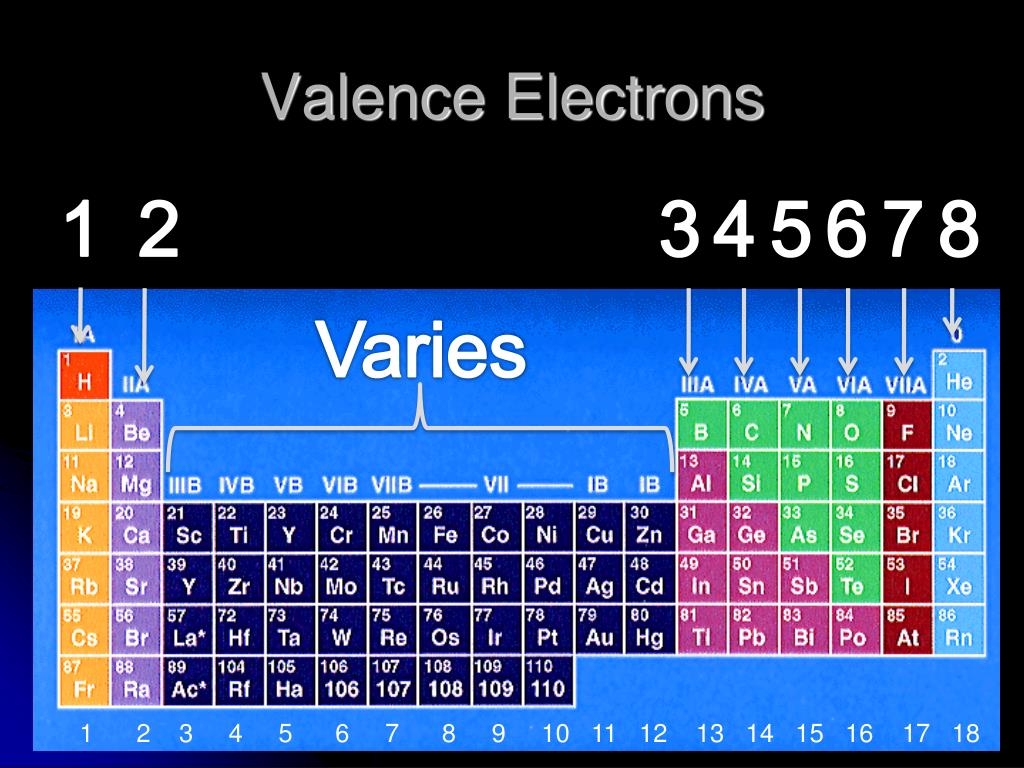
So these are the remaining electrons that are not valence electrons. Now, besides the valence electrons, all the remaining electrons are called your inner core electrons. Now these valence electrons are the outer shell electrons involved in forming chemical bonds. Now for main group elements were going to say that the number of valence electrons that they possess equals their group number.
